Subutex: What is Subutex? (With Photos)
What is Subutex?
The Subutex brand was discontinued in 2011, but the word is used to describe buprenorphine-only medication, as opposed to Suboxone which also contains naloxone for a safeguard.
Both Subutex and Suboxone treat opioid addiction by keeping cravings and withdrawal symptoms manageable.
Skip To
Subutex vs. Suboxone | Discontinued | Subutex Tablets | Subutex Side Effects | FAQ | Getting Subutex | Ask a Question
Subutex vs Suboxone
Both Subutex and Suboxone have the same active ingredient — buprenorphine. But, Suboxone has an additional medication called naloxone that isn’t found in Subutex.
Naloxone doesn’t impact the function of Suboxone unless it is abused. If Suboxone is taken in excess or injected, the Naloxone can send a person into withdrawal instead of giving off a high and protects against overdose death. Subutex doesn’t have this safeguard.
Suboxone is safer than Subutex (buprenorphine-only).
| Compare | Suboxone | Subutex |
| Ingredients | Buprenorphine + Naloxone | Buprenorphine |
| Includes Safeguard Against Abuse | Yes | No |
| Delivery | Sublingual film under tongue | Sublingual tablet under tongue or monthly injection |
| Brand Names | Suboxone, Zubsolv, generic | Subutex (discontinued), Sublocade (monthly injection), Belbuca, generic |
| Most Common Uses | Outpatient treatment / at-home prescription For opioid detox & ongoing maintenance | Inpatient treatment For opioid detox & maintenance usually only if pregnant or Naloxone allergies |
Subutex for Detox
Some addiction treatment providers prescribe Subutex (buprenorphine only) for the first days of opioid detox, then switch patients to Suboxone (buprenorphine + naloxone).
The idea is to reduce the risk of precipitated withdrawal by not including naloxone. However, both Subutex and Suboxone cause precipitated withdrawals when induction dosages are not timed correctly. Working with a doctor experienced in opioid detox is the best way to avoid precipitated withdrawals. Suboxone is the safer medication for outpatient use. ⓘ
Why Was Subutex Brand Discontinued?
Subutex was developed by the same manufacturer as Suboxone, now known as Indivior. Indivior cited safer alternatives as the reason for the discontinuation of Subutex in 2011.
Generic buprenorphine-only medications are still used in specific situations like inpatient settings or during pregnancy. But, Suboxone is safer and therefore a better option for most patients, especially for at-home use.
What Does Subutex Look Like?
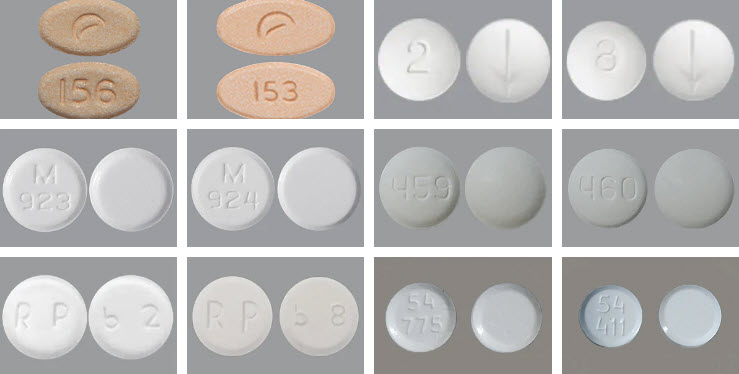
Subutex brand tablets don’t exist anymore, but several manufacturers make buprenorphine-only tablets in 2mg and 8mg doses. These are probably what someone is referring to when they say “Subutex pill”.
Buprenorphine-Only Tablets
2mg 8mg 2mg 8mg 2mg 8mg 2mg 8mg 2mg 8mg 2mg 8mg
While they may look like pills, all are actually taken as tablets that dissolve under your tongue.
Subutex Side Effects
The medication in Subutex (buprenorphine) is well-tolerated.
The most common side effect reported by Symetria patients is constipation. Over-the-counter laxatives, fiber supplements or diet changes can help.
Less Common Side Effects of Subutex
- Drowsiness (shouldn’t occur when stabilized on the right dose)
- Nausea / Vomiting
- Headache
- Sleep issues (including insomnia)
- Dental issues (including tooth decay and infections) ⓘ
Precipitated Withdrawal
Precipitated withdrawals are opioid withdrawal symptoms that hit patients suddenly after taking an anti-craving medication like Subutex too soon.
Symptoms of precipitated withdrawals can be severe and include diarrhea, headache, vomiting, anxiety, runny nose, body aches and chills.
If your initial dose of Subutex (buprenorphine) is taken at a clinic that’s experienced with opioid withdrawal, the risk of precipitated withdrawal is low and can be treated. Subutex should only be started when already in a state of withdrawal.
Subutex Abuse
Subutex (buprenorphine) has a ceiling effect that limits the euphoria and risk of overdose compared to full opioids like heroin or morphine. Patients with an opioid tolerance aren’t likely to feel “high” with Subutex and taking more Subutex does not give more euphoria.
Full vs. Partial Opioids
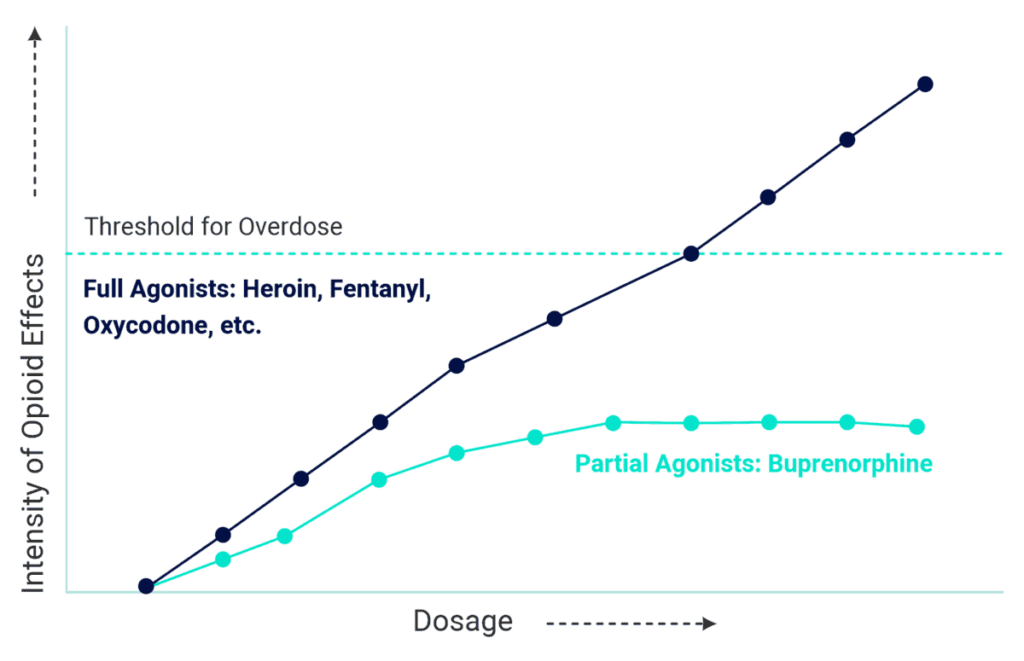
That said, Subutex still carries a higher potential for abuse than Suboxone, which contains the Naloxone safeguard. And, to stop taking Subutex, a buprenorphine taper should be used instead of quitting cold turkey.
Subutex FAQs
What does Subutex do to your body?
Subutex (buprenorphine) partially activates opioid receptors in the brain.
Because buprenorphine satisfies these opioid receptors, they don’t send signals triggering withdrawal symptoms or cravings.
Only partially activating the opioid receptors makes buprenorphine safer than full opioids like heroin, hydrocodone (Vicodin), oxycodone (OxyContin, Percocet), etc. and prevents the same “high”.
Can you switch between Suboxone and Subutex?
Suboxone and Subutex contain the same active ingredient, so switching between the medications is simple. Most patients taking Subutex make the switch to Suboxone, whether it’s after a few days of detox or after pregnancy. There are not many situations where a doctor would agree to switch a patient from Suboxone to Subutex.
Subutex (buprenorphine) is used to treat Opioid Use Disorder but is not a first-choice medication.
Subutex is usually only used if a patient is:
- Pregnant
- Allergic to naloxone
Is Subutex safer than methadone?
Subutex (buprenorphine) is a safer medication than methadone, but methadone has more regulations around prescription and dispensing to help with safety issues. That means both medications are safe treatment options when taken as directed.
See also buprenorphine vs. methadone.
Are there online Subutex doctors?
In response to COVID-19, restrictions were temporarily altered to allow online prescription of buprenorphine. (Though, states like Alabama have rolled back these measures).
However, there are limited situations where a doctor would prescribe Subutex — whether in person or online. Suboxone is the default medication choice because it is safer, which would be even more important in an online setting without having vitals or confirmed drug screen data.
Subutex Doctors Near Me
Subutex brand is no longer available, but buprenorphine-only medication is still used in specific circumstances. And, all patients can get relief from opioid withdrawal symptoms and stop cravings with buprenorphine-based medications.
Subutex Doctors Near Illinois & Texas
Illinois Buprenorphine Clinics
- Subutex Chicago
- Subutex Naperville
- Subutex Joliet
- Subutex Des Plaines
- Subutex Palos Heights
- Subutex Vernon Hills
Texas Buprenorphine Clinics
Ready for Real Recovery — On Your Terms?
Same-day appointments. Full insurance coverage. No judgment.
Fill out the confidential form and take your first step with Symetria today.
DAP PPC forms - Blog 2026
We respond within 1 business day. Your information is 100% confidential and HIPAA compliant.